Think Forward. Then Forward Again.
When tariffs strike and states watch: Life sciences scenario planning for an unpredictable future
The life sciences landscape has a habit of shifting just when you think you've got it figured out. What used to be an industry built on predictable cycles now feels more like navigating through a constant storm of change. We've already explored reimportation challenges and public sector negotiations in scenario planning. Still, there's a fundamental truth worth remembering: the scenarios that catch us off guard are usually the ones that reshape everything.
You're dealing with forces that seemed unimaginable just a few years ago. The good news? Effective scenario planning isn't about becoming a fortune teller — it's about building the organizational muscle to respond when the future inevitably throws you a curveball.
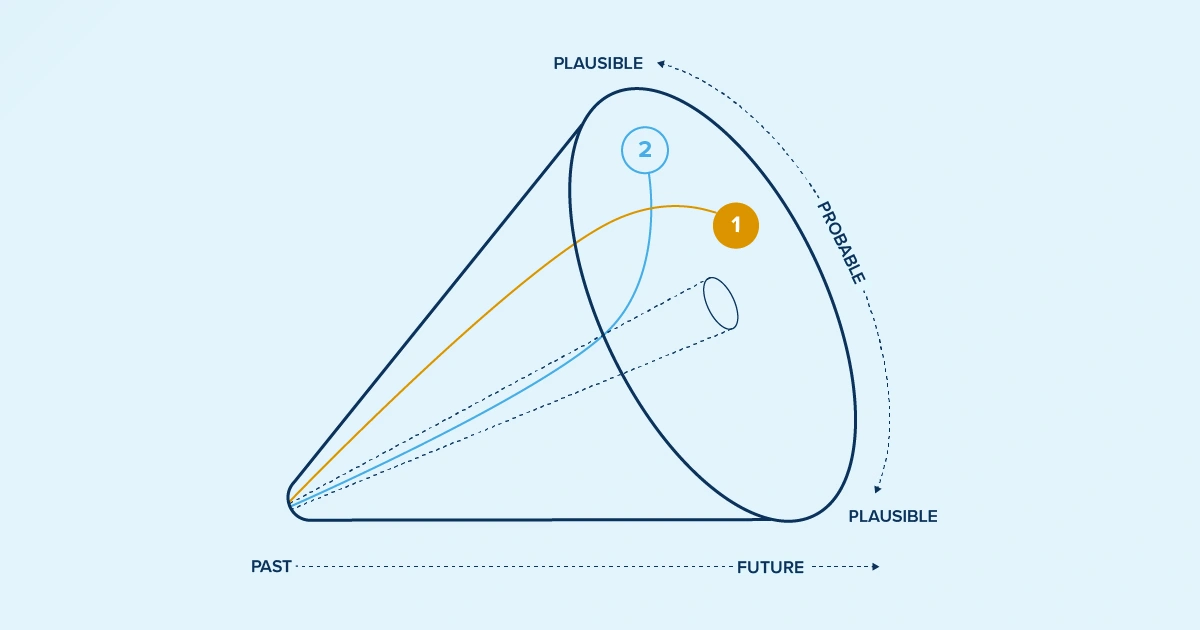
Let's explore two emerging life sciences scenarios that are reshaping how you need to think about pricing, market access and reimbursement. These aren't distant possibilities on the horizon; they're realities that smart life sciences manufacturers are already grappling with.
Scenario 1: Tariff unpredictability and global supply chain disruption
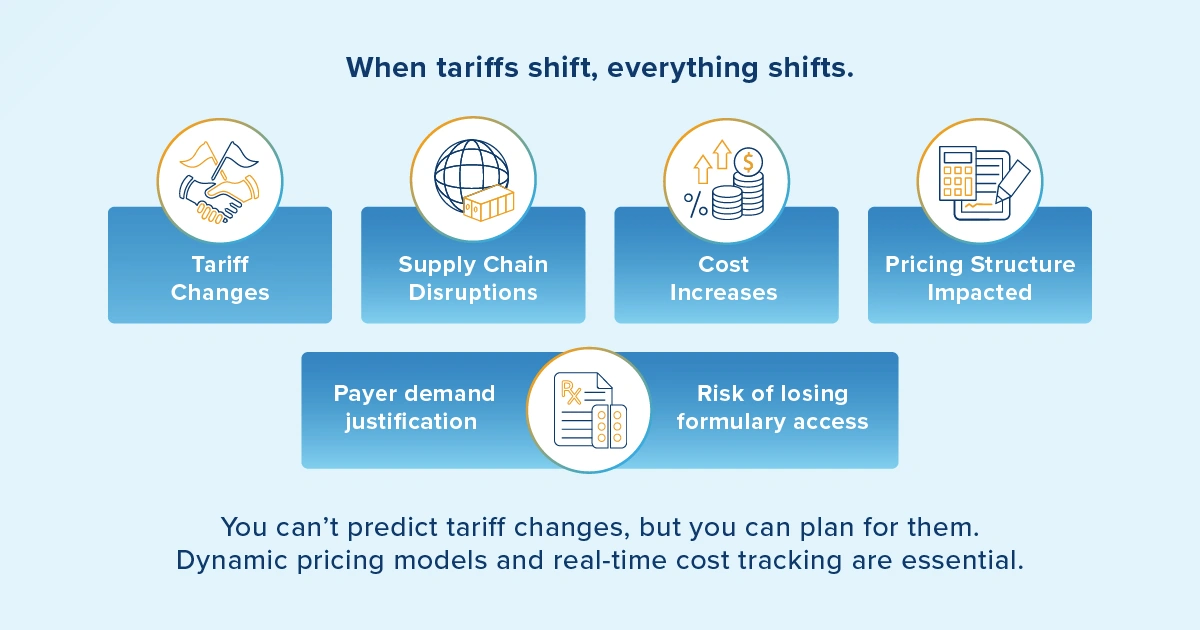
The interconnected nature of pharmaceutical manufacturing and supply chains has created vulnerabilities that will fundamentally alter how you approach pricing and reimbursement strategies. Pandemics and conflicts can create global supply issues. Tariff unpredictability has now emerged as a critical concern that extends far beyond simple cost considerations, and for good reason.
Your industry relies heavily on global supply chains, with active pharmaceutical ingredients (APIs) sourced from multiple countries and manufacturing facilities spanning continents. Trade policy shifts can introduce sudden cost volatility that ripples through every aspect of your business model. What makes this scenario particularly challenging is its unpredictability — tariffs can be implemented with little warning, creating immediate pressure on established pricing structures.
From a reimbursement perspective, this scenario requires you to consider how payers will respond to your claims for increased costs resulting from tariffs. Will they accept these explanations as justification for higher prices, or will they demand that you absorb these costs to maintain formulary access?
Here's the reality: payers are becoming increasingly skeptical of external cost pressures, viewing them as manufacturers' problems rather than shared market challenges.
The long-term solution is to spread manufacturing capacity globally and expand supply chain options. This will, of course, require time and investment. To mitigate costs, you may also consider forming consortia to share investments in global capacity and supply chain expansion.
The implications for scenario planning are significant. In the near term, you need dynamic pricing models that can quickly adjust to supply chain disruptions and sudden shifts in tariffs, while maintaining a competitive position. This requires advanced data management systems that can track cost variations across multiple suppliers and regions — exactly the kind of complex data environment where Vistex excels in maintaining pricing accuracy and ensuring compliance.
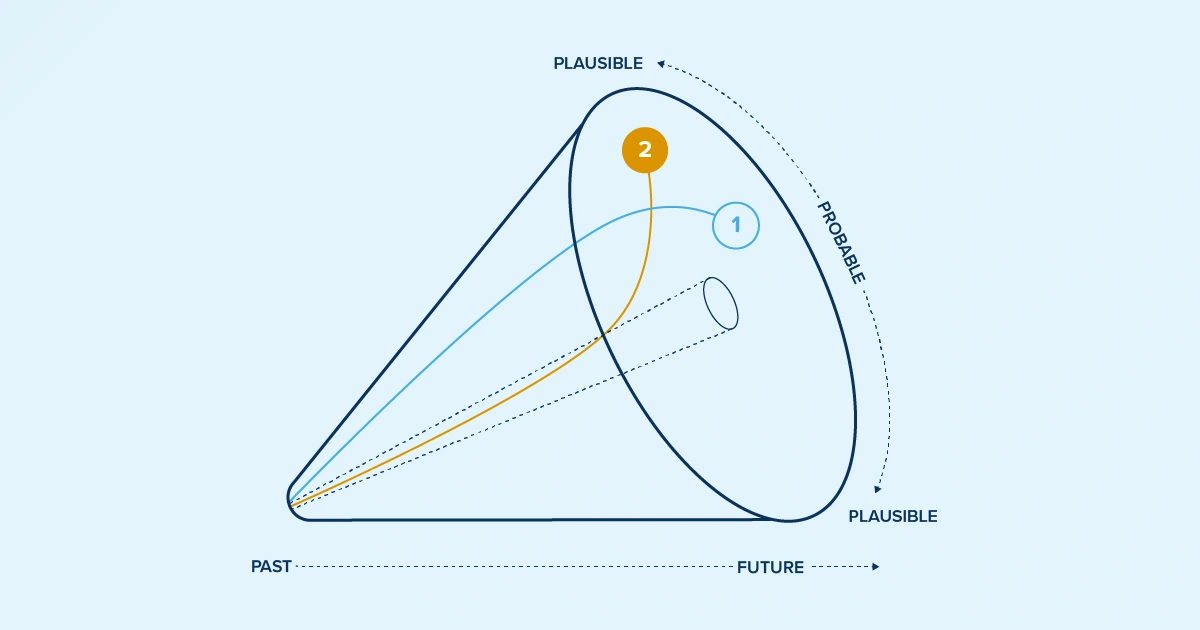
Scenario 2: Price transparency and increasing state control
State price transparency initiatives are gaining momentum. While the stringency varies significantly — with some states requiring comprehensive reporting and others taking more limited approaches, the trajectory is clear: this patchwork of regulations is evolving into a more comprehensive oversight system.
State governments are no longer content to wait for federal action on drug pricing. They're implementing their own reporting requirements and price monitoring systems. What started as simple transparency reporting has evolved into sophisticated oversight programs that can influence market dynamics in real-time.
The states are increasingly examining all stages and players in the value chain and assessing their impact on the cost of pharmaceuticals and healthcare. You will need to adapt to potential changes in the marketplace now, as some or all players in the value chain may face increased regulation.
This scenario presents unique challenges because it fragments what was once a relatively unified regulatory environment. You might face different reporting requirements, transparency standards and pressures in each state where you operate. The administrative burden alone is staggering, but the strategic implications are even more significant.
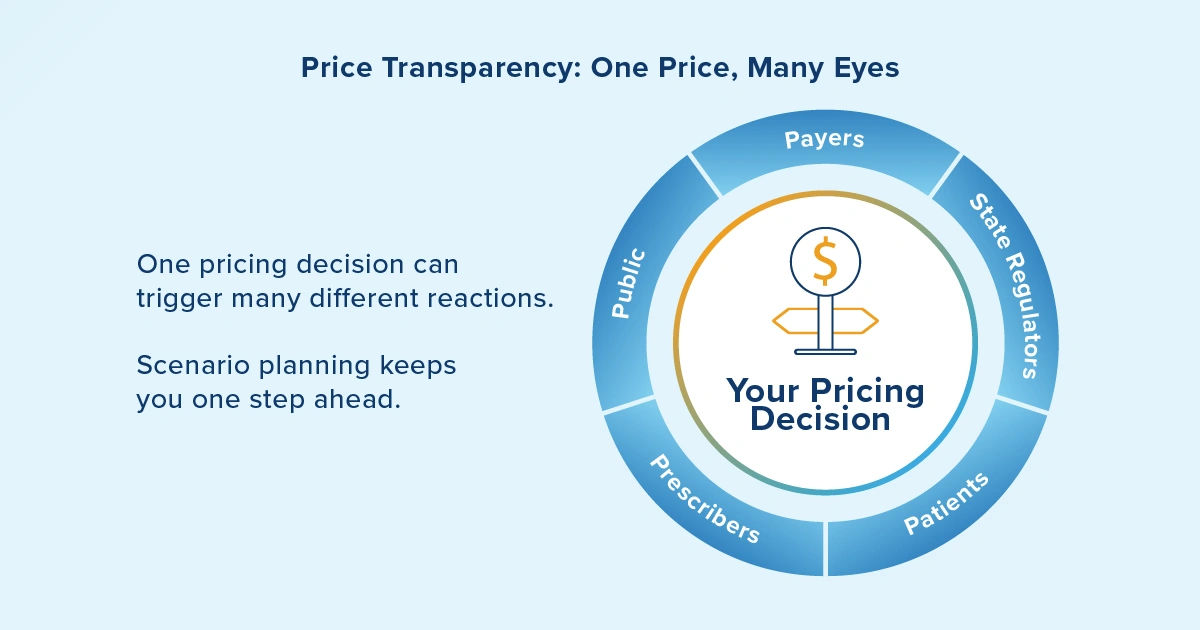
State price transparency creates multiple audiences for your pricing decisions. You must now consider not just payers and patients, but also state regulators, advocacy groups and the public who have unprecedented access to pricing information. This transparency can influence prescribing patterns, formulary decisions and public policy in ways that traditional market research couldn't predict.
The data management requirements for this scenario are complex. You need systems that can track and report pricing information across multiple state jurisdictions, each with potentially different requirements and timelines. You also need the analytical capability to understand how price transparency in one state might influence market dynamics in another.
This will require the ability to run multiple scenarios for each decision point and adapt your models to continuous change. Vistex helps you stay ahead of these shifts by centralizing pricing data, streamlining multi-state reporting and delivering the analytics needed to anticipate how transparency regulations impact access and profitability across the value chain.
Every pricing decision you make tomorrow depends on the scenario planning you do today
These scenarios remind me that the only constant in our industry is change itself. The companies that thrive will be those that embrace scenario planning not as an academic exercise, but as a core business capability. I've seen firsthand how effective use of Vistex scenario planning tools can transform an organization's ability to navigate uncertainty. Invest in the systems, processes and partnerships necessary to respond quickly when the unexpected becomes reality.